E.Z.N.A.® Tissue DNA Kit
$0.00 – $942.90Price range: $0.00 through $942.90
Isolate DNA from tissues, buccal swabs, cultured cells, whole blood, body fluids, paraffin-embedded tissues and mouse tail snips using mini spin columns
- Rapid – DNA isolation in less than 20 minutes post-lysis
- Versatile – Single kit for multiple sample types
- Specialized buffer system – Optimized buffers for higher yields
- Safe – No phenol/chloroform extractions
- High-quality – DNA is suitable for a variety of downstream applications
The E.Z.N.A. Tissue DNA Kit offers a versatile and cost-effective method for isolating DNA from a wide variety of samples, including fresh or frozen animal cultured cells and tissues, buccal swabs, whole blood, mouse tail snips, etc. The DNA purification process is simplified with Silica Mini Spin Column technology into four quick lyse, bind, wash, and elute steps and can be accomplished in less than 20 minutes post-lysis. This convenient spin-column format avoids time-consuming steps like alcohol precipitation and the use of toxic compounds such as phenol and chloroform and allows for multiple samples to be processed in parallel. DNA purified using this kit is ready for most downstream applications such as PCR, sequencing, genotyping, southern blot analysis, and restriction enzyme digestion.
For Research Use Only. Not for use in diagnostic procedures.
| FEATURES | SPECIFICATIONS |
|---|---|
| Downstream Applications | PCR, sequencing, genotyping, southern blot analysis and restriction enzyme digestion |
| Starting Material | Tissues, cultured cells, mouse tail snips, paraffin-embedded tissues, whole blood, body fluids, buccal swabs |
| Starting Amount | 30 mg, or 5 x 106 cultured cells |
| Elution Volume | 100-200 μL |
| DNA Binding Technology | Silica mini spin column |
| Throughput | 1-24 |
| Processing Time | < 20 min (post-lysis) |
| ITEM | AVAILABLE SEPARATELY |
|---|---|
| HiBind® DNA Mini Columns | View Product |
| 2 mL Collection Tubes | View Product |
| BL Buffer | View Product |
| TL Buffer | View Product |
| HBC Buffer | View Product |
| DNA Wash Buffer | View Product |
| Elution Buffer | View Product |
| Proteinase K Solution | View Product |
Higher Yields of High-Quality DNA Extracted from Cells Compared to Company Q

Figure 1. Genomic DNA was isolated from 1 x 106 AD293 cells with Omega Bio-tek’s E.Z.N.A.® Tissue DNA Kit, as well as an equivalent kit from Company Q. DNA was eluted in 200 µL . DNA concentration was determined by optical density measurements using Thermo Scientific’s NanoDrop 2000c system. Omega Bio-tek’s Kit extracted higher yields of high-quality DNA compared to Company Q’s kit.
Unfragmented DNA Extracted From Cells Using Omega Bio-tek’s Kit
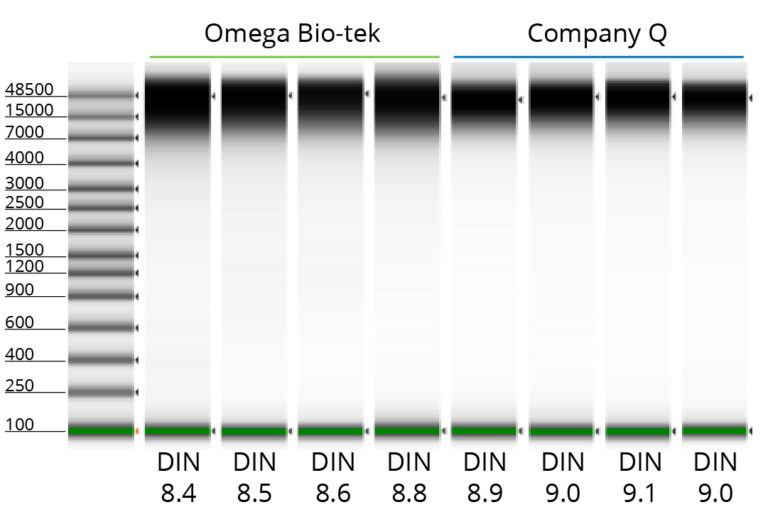
Figure 2. Genomic DNA was isolated using 1 x 106 AD293 cells with Omega Bio-tek’s E.Z.N.A.® Tissue DNA Kit, as well as an equivalent kit from Company Q. DNA was eluted in 200 µL . Electrophoretic separation and analysis were performed through Agilent’s TapeStation® 4150 to determine DNA concentration and integrity. DNA integrity was comparable between kits.
Inhibitor-Free DNA Extraction from Cells

Figure 3. Genomic DNA was isolated using 1 x 106 AD293 cells with Omega Bio-tek’s E.Z.N.A.® Tissue DNA Kit, as well as an equivalent kit from Company Q. The extracted DNA was subjected to qPCR to evaluate its suitability for downstream processes. The ΔCt for the extracted genomic DNA using Omega Bio-tek’s Kit was ~3.3, indicating no PCR inhibition. Ct values were acquired using Applied Biosystems’ QuantStudio™ 3 Real-Time PCR System.
Higher Yields of High-Quality DNA Extracted from Muscle Tissue Compared to Company Q
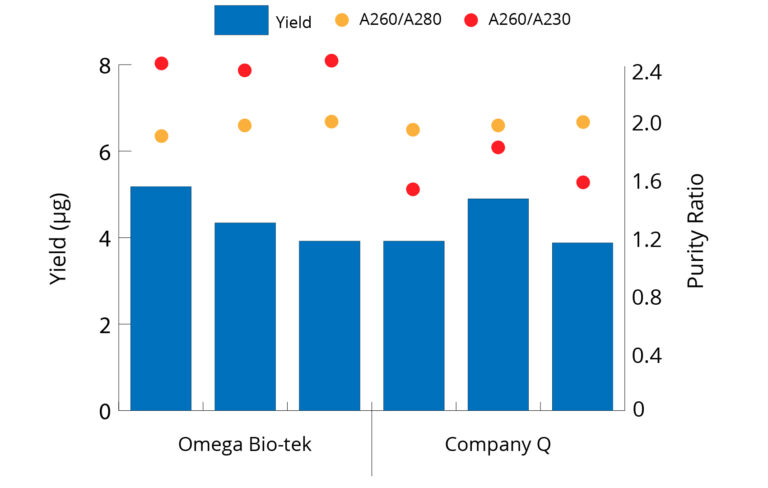
Figure 4. Genomic DNA was isolated from 22-25 mg of goat muscle (n=3) using Omega Bio-tek’s E.Z.N.A.® Tissue DNA Kit, as well as an equivalent kit from Company Q. DNA was eluted in 200 µL . DNA concentration was determined by optical density measurements using Thermo Scientific’s NanoDrop™ 2000c system. DNA integrity was comparable between kits.
Unfragmented DNA Extraction from Muscle Tissues Using Omega Bio-tek's Kit
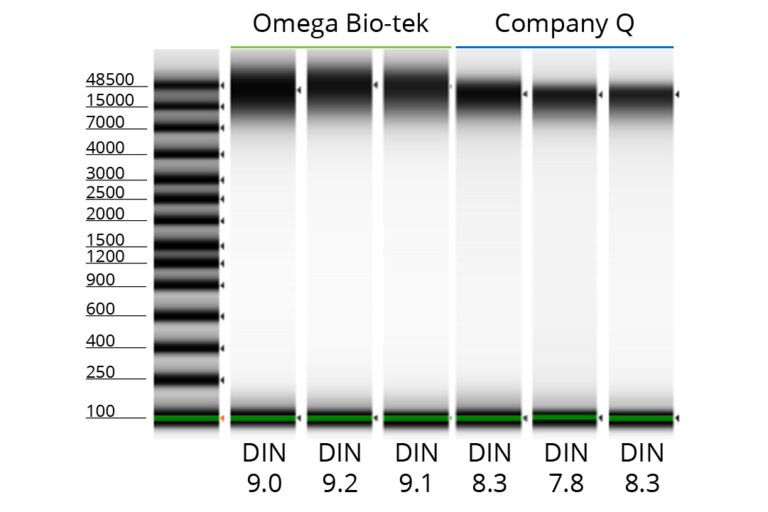
Figure 5. Genomic DNA was isolated from 22-25 mg of goat muscle (n=3) using Omega Bio-tek’s E.Z.N.A.® Tissue DNA Kit, as well as an equivalent kit from Company Q. DNA was eluted in 200 µL. Electrophoretic separation and analysis were performed through Agilent’s TapeStation® 4150 to determine DNA concentration and integrity.
Inhibitor-Free DNA Extraction
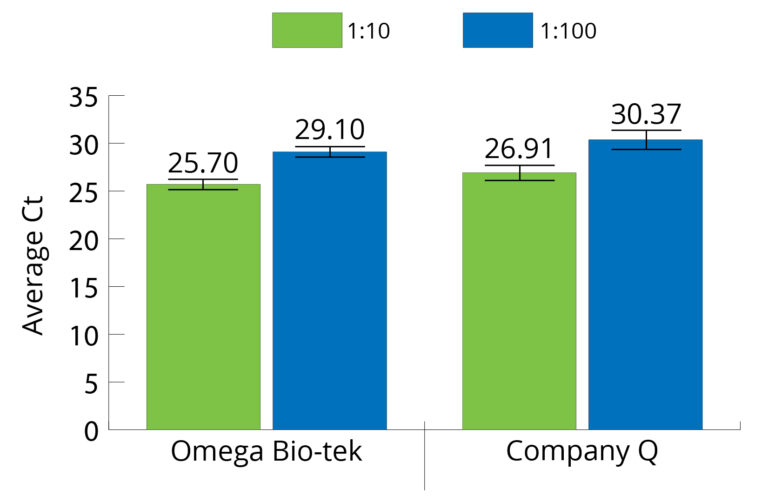
Figure 6. Genomic DNA was isolated from 22-25 mg of goat muscle (n=3) using Omega Bio-tek’s E.Z.N.A.® Tissue DNA Kit, as well as an equivalent kit from Company Q. The extracted DNA was subjected to qPCR to evaluate its suitability for downstream processes. The ΔCt for the extracted genomic DNA was ~3.3, indicating no PCR inhibition. Ct values were acquired using Applied Biosystems’ QuantStudio™ 3 Real-Time PCR System.
FAQs
Can the E.Z.N.A. Tissue DNA Kit extract DNA from hair?
Yes. For hair, please add DTT to a final concentration of 40 mM to the lysis buffer (TL) and extend incubation at 55 °C as needed.
What extraction kit do you recommend for extracting DNA from yeast?
We recommend D3396 with protocol modifications.
The modification will require making or sourcing Sorbitol buffer (1 M sorbitol; 100 mM sodium EDTA; 14 mM β-mercaptoethanol) and obtaining lyticase. Before beginning the tissue DNA protocol on page 8 of the E.Z.N.A. Tissue DNA Kit, you will need to perform the steps outlined below.
- Harvest cells (maximum 2 x 107) by centrifuging for 10 minutes at 5,000 x g (approximately 7,500 rpm). Discard the supernatant.
- Resuspend the pellet in 600 µL sorbitol buffer and 200 units of lyticase. Incubate at 30 °C for 30 minutes.
- Pellet the spheroplasts by centrifuging for 10 minutes at 300 x g.
Continue on using step 2 of the Tissue DNA protocol found on page 8 of the E.Z.N.A. Tissue DNA Kit product manual.
Product Information
Safety Data Sheets
| Components | Hazard Standards | Languages | Link | hf:tax:dlp_document_language | hf:tax:dlp_document_hazard-standard |
|---|---|---|---|---|---|
| BL Buffer | GHS | English | english | ghs | |
| BL Buffer | GHS | Spanish | spanish | ghs | |
| BL Buffer | REACH | English | english | reach | |
| BL Buffer | REACH | Danish | danish | reach | |
| BL Buffer | REACH | Finnish | finnish | reach | |
| BL Buffer | REACH | French | french | reach | |
| BL Buffer | REACH | German | german | reach | |
| BL Buffer | REACH | Italian | italian | reach | |
| BL Buffer | REACH | Norwegian | norwegian | reach | |
| BL Buffer | REACH | Spanish | spanish | reach | |
| BL Buffer | REACH | Swedish | swedish | reach | |
| BL Buffer | WHMS | English | english | whms | |
| BL Buffer | WHMS | French | french | whms | |
| BL Buffer | REACH | Latvian | latvian | reach | |
| BL Buffer | REACH | Polish | polish | reach | |
| BL Buffer | REACH | Portuguese | portuguese | reach | |
| BL Buffer | REACH | Greek | greek | reach | |
| DNA Wash Buffer | GHS | English | english | ghs | |
| DNA Wash Buffer | GHS | Spanish | spanish | ghs | |
| DNA Wash Buffer | REACH | English | english | reach | |
| DNA Wash Buffer | REACH | Danish | danish | reach | |
| DNA Wash Buffer | REACH | Finnish | finnish | reach | |
| DNA Wash Buffer | REACH | French | french | reach | |
| DNA Wash Buffer | REACH | German | german | reach | |
| DNA Wash Buffer | REACH | Italian | italian | reach | |
| DNA Wash Buffer | REACH | Norwegian | norwegian | reach | |
| DNA Wash Buffer | REACH | Spanish | spanish | reach | |
| DNA Wash Buffer | REACH | Swedish | swedish | reach | |
| DNA Wash Buffer | WHMS | English | english | whms | |
| DNA Wash Buffer | WHMS | French | french | whms | |
| DNA Wash Buffer | REACH | Latvian | latvian | reach | |
| DNA Wash Buffer | REACH | Polish | polish | reach | |
| Elution Buffer | GHS | English | english | ghs | |
| Elution Buffer | GHS | Spanish | spanish | ghs | |
| Elution Buffer | REACH | English | english | reach | |
| Elution Buffer | REACH | Danish | danish | reach | |
| Elution Buffer | REACH | Finnish | finnish | reach | |
| Elution Buffer | REACH | French | french | reach | |
| Elution Buffer | REACH | German | german | reach | |
| Elution Buffer | REACH | Italian | italian | reach | |
| Elution Buffer | REACH | Norwegian | norwegian | reach | |
| Elution Buffer | REACH | Spanish | spanish | reach | |
| Elution Buffer | REACH | Swedish | swedish | reach | |
| Elution Buffer | WHMS | English | english | whms | |
| Elution Buffer | WHMS | French | french | whms | |
| Elution Buffer | REACH | Dutch | dutch | reach | |
| Elution Buffer | REACH | Hungarian | hungarian | reach | |
| Elution Buffer | REACH | Portuguese | portuguese | reach | |
| Elution Buffer | REACH | Greek | greek | reach | |
| Elution Buffer | REACH | Latvian | latvian | reach | |
| Elution Buffer | REACH | Polish | polish | reach | |
| HBC Buffer | GHS | English | english | ghs | |
| HBC Buffer | GHS | Spanish | spanish | ghs | |
| HBC Buffer | REACH | English | english | reach | |
| HBC Buffer | REACH | Danish | danish | reach | |
| HBC Buffer | REACH | Finnish | finnish | reach | |
| HBC Buffer | REACH | French | french | reach | |
| HBC Buffer | REACH | German | german | reach | |
| HBC Buffer | REACH | Italian | italian | reach | |
| HBC Buffer | REACH | Norwegian | norwegian | reach | |
| HBC Buffer | REACH | Spanish | spanish | reach | |
| HBC Buffer | REACH | Swedish | swedish | reach | |
| HBC Buffer | WHMS | English | english | whms | |
| HBC Buffer | WHMS | French | french | whms | |
| HBC Buffer | REACH | Latvian | latvian | reach | |
| HBC Buffer | REACH | Polish | polish | reach | |
| Proteinase K Solution | GHS | English | english | ghs | |
| Proteinase K Solution | GHS | Spanish | spanish | ghs | |
| Proteinase K Solution | REACH | English | english | reach | |
| Proteinase K Solution | REACH | Danish | danish | reach | |
| Proteinase K Solution | REACH | Finnish | finnish | reach | |
| Proteinase K Solution | REACH | French | french | reach | |
| Proteinase K Solution | REACH | German | german | reach | |
| Proteinase K Solution | REACH | Italian | italian | reach | |
| Proteinase K Solution | REACH | Norwegian | norwegian | reach | |
| Proteinase K Solution | REACH | Spanish | spanish | reach | |
| Proteinase K Solution | REACH | Swedish | swedish | reach | |
| Proteinase K Solution | WHMS | English | english | whms | |
| Proteinase K Solution | WHMS | French | french | whms | |
| Proteinase K Solution | REACH | Dutch | dutch | reach | |
| Proteinase K Solution | REACH | Hungarian | hungarian | reach | |
| Proteinase K Solution | REACH | Portuguese | portuguese | reach | |
| Proteinase K Solution | REACH | Greek | greek | reach | |
| Proteinase K Solution | REACH | Latvian | latvian | reach | |
| Proteinase K Solution | REACH | Polish | polish | reach | |
| TL Buffer | GHS | English | english | ghs | |
| TL Buffer | GHS | Spanish | spanish | ghs | |
| TL Buffer | REACH | English | english | reach | |
| TL Buffer | REACH | Danish | danish | reach | |
| TL Buffer | REACH | Finnish | finnish | reach | |
| TL Buffer | REACH | French | french | reach | |
| TL Buffer | REACH | German | german | reach | |
| TL Buffer | REACH | Italian | italian | reach | |
| TL Buffer | REACH | Norwegian | norwegian | reach | |
| TL Buffer | REACH | Spanish | spanish | reach | |
| TL Buffer | REACH | Swedish | swedish | reach | |
| TL Buffer | WHMS | English | english | whms | |
| TL Buffer | WHMS | French | french | whms | |
| TL Buffer | REACH | Dutch | dutch | reach | |
| TL Buffer | REACH | Hungarian | hungarian | reach | |
| TL Buffer | REACH | Portuguese | portuguese | reach | |
| TL Buffer | REACH | Greek | greek | reach | |
| TL Buffer | REACH | Latvian | latvian | reach | |
| TL Buffer | REACH | Polish | polish | reach |

