The E.Z.N.A.® Endo-Free Plasmid DNA Midi Kit provides an efficient method for the large-scale isolation of low-endotoxin plasmid DNA. The system utilizes Lysate Clearance Filter Syringes instead of centrifugation to rapidly clear bacterial lysates post-alkaline lysis. Following lysate clearance, endotoxins are removed, and the protocol follows a simple “bind-wash-elute” procedure to deliver high-quality plasmid DNA. The yields vary according to plasmid copy number, E. coli strain, and growth conditions. 15-50 mL of bacterial cultures in LB medium typically produces up to 200 µg of high copy number plasmid DNA. Up to 100 mL of culture may be processed when working with low copy number plasmid. The system uses centrifugation or vacuum technology for plasmid purification and eliminates the time-consuming gravity-flow columns that require alcohol precipitation. Purified plasmid DNA is suitable for sensitive downstream applications such as transfection of mammalian cells, cloning, automated fluorescent DNA sequencing (typical reads exceed 800 bp), next-generation sequencing, restriction enzyme digestion, ligation, PCR, in vitro transcription, transformation, etc.
For Research Use Only. Not for use in diagnostic procedures.
| FEATURES | SPECIFICATIONS |
|---|---|
| Downstream Application | Transfection, gene therapy, cloning, in vitro Transcription, Next-Generation Sequencing, PCR, Transformation, restriction digestion, ligation |
| Starting material | 15-50 mL LB culture with OD600 between 2 and 3; or equivalent |
| Plasmid type | High-copy, low-copy, cosmid DNA |
| Processing mode | Manual (centrifugation or vacuum) |
| Throughput | 1 – 24 |
| DNA binding technology | Silica Midi Spin Column |
| Lysate clearance method | Filtration using Lysate clearance syringe |
| Processing time | < 45 minutes |
| Yield | 100-250 µg for high copy-number; 10-50 µg for low copy-number |
| Special note | Includes endotoxin removal step |
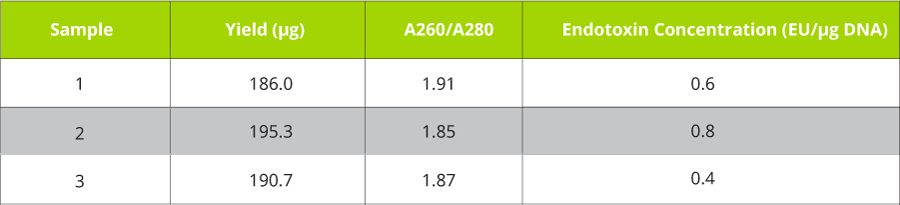
Table 1. pEGFP-N1 plasmid was purified from 50 mL DH5α cultures harboring the plasmid using Omega Bio-tek’s E.Z.N.A.® Endo-Free Plasmid DNA Midi Kit following manufacturer’s recommended protocol. Plasmid DNA was eluted in 500 µL volume and quantified using Thermo Scientific’s NanoDrop™ 2000c system. The Endotoxin concentration was estimated based on a Limulus Amoebocyte Lysate (LAL) assay.
Product Information
| Document Name | Languages | Link |
|---|---|---|
| Protocol Manual | English |
Safety Data Sheets
| Components | Hazard Standards | Languages | Link | hf:tax:dlp_document_language | hf:tax:dlp_document_hazard-standard |
|---|---|---|---|---|---|
| DNA Wash Buffer | GHS | English | english | ghs | |
| DNA Wash Buffer | GHS | Spanish | spanish | ghs | |
| DNA Wash Buffer | REACH | English | english | reach | |
| DNA Wash Buffer | REACH | Danish | danish | reach | |
| DNA Wash Buffer | REACH | Finnish | finnish | reach | |
| DNA Wash Buffer | REACH | French | french | reach | |
| DNA Wash Buffer | REACH | German | german | reach | |
| DNA Wash Buffer | REACH | Italian | italian | reach | |
| DNA Wash Buffer | REACH | Norwegian | norwegian | reach | |
| DNA Wash Buffer | REACH | Spanish | spanish | reach | |
| DNA Wash Buffer | REACH | Swedish | swedish | reach | |
| DNA Wash Buffer | WHMS | English | english | whms | |
| DNA Wash Buffer | WHMS | French | french | whms | |
| DNA Wash Buffer | REACH | Latvian | latvian | reach | |
| DNA Wash Buffer | REACH | Polish | polish | reach | |
| EHBC Buffer | GHS | English | english | ghs | |
| EHBC Buffer | GHS | Spanish | spanish | ghs | |
| EHBC Buffer | REACH | English | english | reach | |
| EHBC Buffer | REACH | Danish | danish | reach | |
| EHBC Buffer | REACH | Finnish | finnish | reach | |
| EHBC Buffer | REACH | French | french | reach | |
| EHBC Buffer | REACH | German | german | reach | |
| EHBC Buffer | REACH | Italian | italian | reach | |
| EHBC Buffer | REACH | Norwegian | norwegian | reach | |
| EHBC Buffer | REACH | Spanish | spanish | reach | |
| EHBC Buffer | REACH | Swedish | swedish | reach | |
| EHBC Buffer | WHMS | English | english | whms | |
| EHBC Buffer | WHMS | French | french | whms | |
| EHBC Buffer | REACH | Latvian | latvian | reach | |
| EHBC Buffer | REACH | Polish | polish | reach | |
| Endo-Free Elution Buffer | GHS | English | english | ghs | |
| Endo-Free Elution Buffer | GHS | Spanish | spanish | ghs | |
| Endo-Free Elution Buffer | REACH | English | english | reach | |
| Endo-Free Elution Buffer | REACH | Danish | danish | reach | |
| Endo-Free Elution Buffer | REACH | Finnish | finnish | reach | |
| Endo-Free Elution Buffer | REACH | French | french | reach | |
| Endo-Free Elution Buffer | REACH | German | german | reach | |
| Endo-Free Elution Buffer | REACH | Italian | italian | reach | |
| Endo-Free Elution Buffer | REACH | Norwegian | norwegian | reach | |
| Endo-Free Elution Buffer | REACH | Spanish | spanish | reach | |
| Endo-Free Elution Buffer | REACH | Swedish | swedish | reach | |
| Endo-Free Elution Buffer | WHMS | English | english | whms | |
| Endo-Free Elution Buffer | WHMS | French | french | whms | |
| Endo-Free Elution Buffer | REACH | Latvian | latvian | reach | |
| Endo-Free Elution Buffer | REACH | Polish | polish | reach | |
| ETR Binding Buffer | GHS | English | english | ghs | |
| ETR Binding Buffer | GHS | Spanish | spanish | ghs | |
| ETR Binding Buffer | REACH | English | english | reach | |
| ETR Binding Buffer | REACH | Danish | danish | reach | |
| ETR Binding Buffer | REACH | Finnish | finnish | reach | |
| ETR Binding Buffer | REACH | French | french | reach | |
| ETR Binding Buffer | REACH | German | german | reach | |
| ETR Binding Buffer | REACH | Italian | italian | reach | |
| ETR Binding Buffer | REACH | Norwegian | norwegian | reach | |
| ETR Binding Buffer | REACH | Spanish | spanish | reach | |
| ETR Binding Buffer | REACH | Swedish | swedish | reach | |
| ETR Binding Buffer | WHMS | English | english | whms | |
| ETR Binding Buffer | WHMS | French | french | whms | |
| ETR Binding Buffer | REACH | Latvian | latvian | reach | |
| ETR Binding Buffer | REACH | Polish | polish | reach | |
| ETR Wash Buffer | GHS | English | english | ghs | |
| ETR Wash Buffer | GHS | Spanish | spanish | ghs | |
| ETR Wash Buffer | REACH | English | english | reach | |
| ETR Wash Buffer | REACH | Danish | danish | reach | |
| ETR Wash Buffer | REACH | Finnish | finnish | reach | |
| ETR Wash Buffer | REACH | French | french | reach | |
| ETR Wash Buffer | REACH | German | german | reach | |
| ETR Wash Buffer | REACH | Italian | italian | reach | |
| ETR Wash Buffer | REACH | Norwegian | norwegian | reach | |
| ETR Wash Buffer | REACH | Spanish | spanish | reach | |
| ETR Wash Buffer | REACH | Swedish | swedish | reach | |
| ETR Wash Buffer | WHMS | English | english | whms | |
| ETR Wash Buffer | WHMS | French | french | whms | |
| ETR Wash Buffer | REACH | Latvian | latvian | reach | |
| ETR Wash Buffer | REACH | Polish | polish | reach | |
| N3 Buffer | GHS | English | english | ghs | |
| N3 Buffer | GHS | Spanish | spanish | ghs | |
| N3 Buffer | REACH | English | english | reach | |
| N3 Buffer | REACH | Danish | danish | reach | |
| N3 Buffer | REACH | Finnish | finnish | reach | |
| N3 Buffer | REACH | French | french | reach | |
| N3 Buffer | REACH | German | german | reach | |
| N3 Buffer | REACH | Italian | italian | reach | |
| N3 Buffer | REACH | Norwegian | norwegian | reach | |
| N3 Buffer | REACH | Spanish | spanish | reach | |
| N3 Buffer | REACH | Swedish | swedish | reach | |
| N3 Buffer | WHMS | English | english | whms | |
| N3 Buffer | WHMS | French | french | whms | |
| N3 Buffer | REACH | Latvian | latvian | reach | |
| N3 Buffer | REACH | Polish | polish | reach | |
| RNase A | GHS | English | english | ghs | |
| RNase A | GHS | Spanish | spanish | ghs | |
| RNase A | REACH | English | english | reach | |
| RNase A | REACH | Danish | danish | reach | |
| RNase A | REACH | Finnish | finnish | reach | |
| RNase A | REACH | French | french | reach | |
| RNase A | REACH | German | german | reach | |
| RNase A | REACH | Italian | italian | reach | |
| RNase A | REACH | Norwegian | norwegian | reach | |
| RNase A | REACH | Spanish | spanish | reach | |
| RNase A | REACH | Swedish | swedish | reach | |
| RNase A | WHMS | English | english | whms | |
| RNase A | WHMS | French | french | whms | |
| RNase A | REACH | Latvian | latvian | reach | |
| RNase A | REACH | Polish | polish | reach | |
| Solution I | GHS | English | english | ghs | |
| Solution I | GHS | Spanish | spanish | ghs | |
| Solution I | REACH | English | english | reach | |
| Solution I | REACH | Danish | danish | reach | |
| Solution I | REACH | Finnish | finnish | reach | |
| Solution I | REACH | French | french | reach | |
| Solution I | REACH | German | german | reach | |
| Solution I | REACH | Italian | italian | reach | |
| Solution I | REACH | Norwegian | norwegian | reach | |
| Solution I | REACH | Spanish | spanish | reach | |
| Solution I | REACH | Swedish | swedish | reach | |
| Solution I | WHMS | English | english | whms | |
| Solution I | WHMS | French | french | whms | |
| Solution I | REACH | Latvian | latvian | reach | |
| Solution I | REACH | Polish | polish | reach | |
| Solution II | GHS | English | english | ghs | |
| Solution II | GHS | Spanish | spanish | ghs | |
| Solution II | REACH | English | english | reach | |
| Solution II | REACH | Danish | danish | reach | |
| Solution II | REACH | Finnish | finnish | reach | |
| Solution II | REACH | French | french | reach | |
| Solution II | REACH | German | german | reach | |
| Solution II | REACH | Italian | italian | reach | |
| Solution II | REACH | Norwegian | norwegian | reach | |
| Solution II | REACH | Spanish | spanish | reach | |
| Solution II | REACH | Swedish | swedish | reach | |
| Solution II | WHMS | English | english | whms | |
| Solution II | WHMS | French | french | whms | |
| Solution II | REACH | Latvian | latvian | reach | |
| Solution II | REACH | Polish | polish | reach |